Micronutrient combination inhibits two key steps of coronavirus (SARS-CoV-2) infection: viral binding to ACE2 receptor and its cellular expression
A. Goc, Ph.D., W. Sumera, M.Sc., Vadim Ivanov, M.D., Ph.D., Aleksandra Niedzwiecki, Ph.D., Matthias Rath, M.D.
Dr. Rath Research Institute, San Jose, CA
INTRODUCTION
The coronavirus pandemic poses an unprecedented challenge to human health as well as global economies. The quest for vaccines and other approaches to this pandemic thus far focuses on synthetic molecules that are – predictably – associated with a variable degree of adverse effects.
RESULTS
Here we report that a composition of specific micronutrients can almost completely block the interaction between the binding site of the coronavirus and its cellular anchor, the ACE2 receptor. Moreover, this micronutrient composition was able to significantly decrease the expression of the ACE2 receptor on human alveolar (lung) epithelial cells by over 90%.
CONCLUSION
This study provides the basis for effective and safe public health strategies based on an optimum intake of micronutrients. It allows people around the world to actively participate in the prevention of coronavirus infections – beyond wearing masks, social distancing, and other defensive measures.
INTRODUCTION
The rapid spread of the current coronavirus pandemic (COVID-19) is threatening global health, debilitating the economies of the world, and challenging the prosperity of future generations.1 During the first half of 2020, COVID-19 has affected over 16 million people and caused more than 660,000 deaths worldwide.2
Sequencing the whole genome of a virus from patient samples3, the Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) was identified, and the disease caused by this virus was named coronavirus disease 2019 (COVID-19)
Another major step towards curbing the spread of this pandemic was the identification of the pathway by which SARS-CoV-2 infects the cells of the human body. Coronaviruses first bind to specific receptors on the surface of cells. Subsequently they enter the cells and are carried via transport vesicles (endosomes), ultimately reaching the cell core (nucleus) where their genetic material is incorporated into the DNA of human body cells.4
Of particular importance is the first step of the infective pathway, namely the binding of the virus to the receptor on the cell surface (Figure 1). On the part of the coronavirus this binding mechanism involves specific Spike proteins anchored on the surface of the virus containing a receptor-binding domain (RBD) that specifically recognizes its ‘docking site’ – a specific receptor on the surface of the body cells. This receptor, called angiotensin-converting enzyme 2 (ACE2), is an integral membrane protein present on many cells throughout the human body with its strong expression in the heart, vascular system, gastrointestinal system, and kidneys, as well as in type II alveolar cells in the lungs.5,6
This binding mechanism is the decisive determinant of viral infectivity and the development of the COVID-19. Moreover, it is also a major target for the development of vaccines and other therapeutics.7,8
While the search for such interventional drugs and vaccines is ongoing, there is a much more direct – and above all safer – way to prevent this fateful interaction of coronaviruses with the ACE2 receptor: to suppress the expression of the ACE2 receptors in human body cells so they are no longer available for the anchoring of coronaviruses.
In a recent pioneering study, we were able to show that a specific composition of micronutrients containing vitamin C, certain minerals, amino acids, and plant extracts was effective in significantly decreasing cellular ACE2 expression in key types of cells targeted by the virus: human lung small airways (alveolar) epithelial cells and human blood vessel (vascular) endothelial cells. These micronutrients were particularly effective in down-regulating the ACE2 receptor expression under inflammatory conditions, which are associated with coronavirus and other infections.9
The remarkable results of this study triggered the question whether micronutrients are also able to affect other mechanisms essential for coronavirus infections, namely the binding between the virus and the ACE2 receptor. Thus, we aimed at blocking viral entry into human cells – the very same disease mechanism that was selected by essentially all current vaccine-based approaches.
If the results of this study were positive, a new, natural and safe approach would become available to mankind to effectively control the current pandemic.
Thus, in the current study we tested the efficacy of another nutrient composition, containing polyphenols and plant components, on both key aspects of SARS- CoV-2 infectivity: blocking the expression of cellular ACE2 receptor and – at the same time – blocking the binding of a specific SARS-CoV-2 Spike protein to the ACE2 receptor on human cells.
METHODS
Cell cultures
Human Small Airways Epithelial Cells (SAEC, purchased from ATCC) were cultured in Airways Epithelial Cells growth medium (ATCC) in plastic flasks at 37oC and 5% CO2. In the experiments SAEC, passage 5-7, were plated to collagen-covered 96 well plastic plates (Corning) in 100 μL growth medium and were grown for 4-7 days to reach confluent layer.
Micronutrient composition
The micronutrient combination used in our experiments was developed at the Dr. Rath Research Institute (San Jose, CA). The test formulation contained: quercetin – 400 mg, cruciferous plant extract – 400 mg, turmeric root extract – 300 mg, green tea extract (80% polyphenols) – 300 mg, and resveratrol – 50 mg.
Cell supplementation
The micronutrient mixture was dissolved in 0.1N HCl according to US Pharmacopeia protocol (USP 2040) and designated as a stock solution. For ACE2 expression experiments SAEC cells were supplemented with indicated doses of the formulation in cell growth medium at 100 μl/well for 3-7 days. Applied nutrient concentrations were expressed in μg/mL.
ACE-2 ELISA assay
Culture plate wells were washed twice with phosphate buffered saline (PBS) and fixed with 3% formaldehyde/0.5% Triton X100/PBS solution for 1h at 4oC, then washed four times with PBS. Subsequently, 200 μl of 1% bovine serum albumin (BSA, Sigma) in PBS was added and plate was incubated at 4oC overnight. Rabbit polyclonal anti ACE2 antibodies (Sigma) were added to 100 μl 1%BSA/PBS for 1.5 h incubation at room temperature (RT). After three wash cycles with 0.1%BSA/PBS the wells were supplied with 100 μl anti-rabbit IgG antibodies conjugated with horse radish peroxidase (HRP, Sigma) for 1h at RT. After three wash cycles with 0.1%BSA/PBS the retained HRP activity was determined by incubation with 100 μl TMB substrate solution (Sigma) for 20 min at RT, followed by the addition of 50 μl of 1N H2SO4, and optical density measurement were conducted at 450 nm with micro plate reader (Molecular Devices). Results are expressed as a percentage of experimental addition-free control (mean +/- SD, n=6). Non-specific control (wells incubated without anti ACE2 antibodies) mean value (n=6) was subtracted from all sample values.
RBD (receptor-binding domain) blocking
This assay was performed using GenScript SARS-CoV-2 surrogate virus neutralization test kit that can detect either antibody or inhibitors that block the interaction between the receptor binding domain (RBD) of the viral Spike protein and the ACE2 cell surface receptor.
All test samples with indicated concentrations and positive and negative controls (provided by the manufacturer) were diluted with the sample dilution buffer with a volume ratio of 1:9. In separate tubes, HRP conjugated RBD was also diluted with the HRP dilution buffer with a volume ratio of 1:99.
Biding/neutralization reaction was performed according to manufacturer’s protocol. Briefly, diluted positive and negative controls as well as the test samples with indicated concentrations were mixed with the diluted HRP-RBD solution with a volume ratio of 1:1 and incubated for 30 minutes at 37OC. Next, 100 µl each of the positive control mixture, negative control mixture, and the test sample mixtures were added to the corresponding wells with immobilized ACE2 receptor and incubated for 15 minutes at 37OC. Subsequently, the plates were washed four times with 260 µl/well of the 1 x wash solution and TMB solution was added to each well (100 µl/well).
Plates were incubated in the dark at room temperature for up to 5 minutes. Next, 50 µl/well of stop solution was added to quench the reaction and the absorbance was measured immediately in plate reader at 450 nm. Experiment was performed three times in duplicates. Data are presented as % of control.
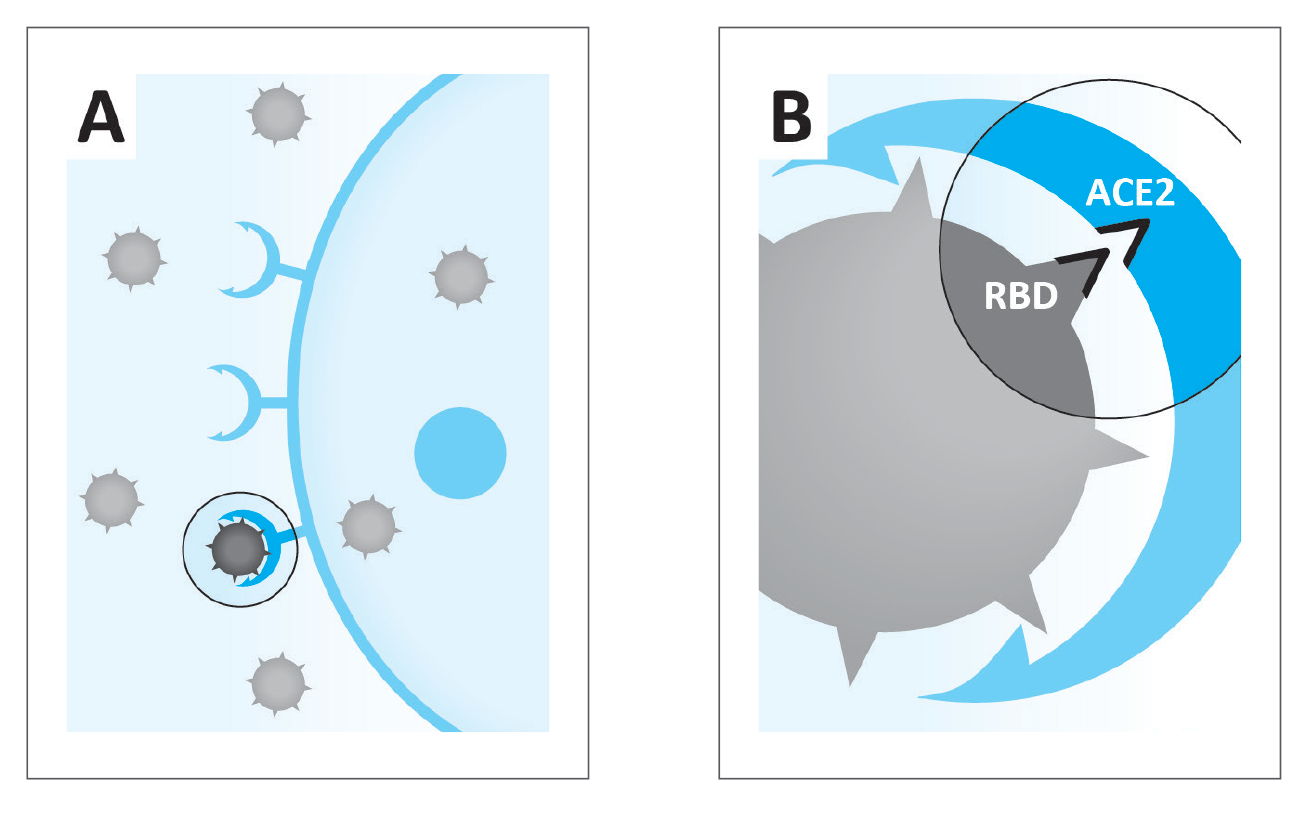 Figure 1:
Figure 1:
A. Coronaviruses infect human body cell via the ACE2
B. Principle of the commercially available research test-kit used in this study to measure interaction between the receptor binding protein (RBD) of the SARS-CoV-2 coronavirus and the ACE2 receptor on the cell
RESULTS
Efficacy of a specific micronutrient combination on ACE2 expression in human small alveolar epithelial cells.
Figure 2 shows the effects of different concentrations of a specific combination of various active plant components and extracts on cellular expression of ACE2 receptors in human small alveolar epithelial cells.
The results show a concentration dependent decrease in cellular ACE2 receptor expression evaluated by a specific antibody binding. At the highest nutrient concentration of 320 ug/mL the expression of cellular ACE2 receptors decreased by 92%. This indicates that in the presence of these micronutrients the viral binding to these cell surface receptors can be substantially reduced.
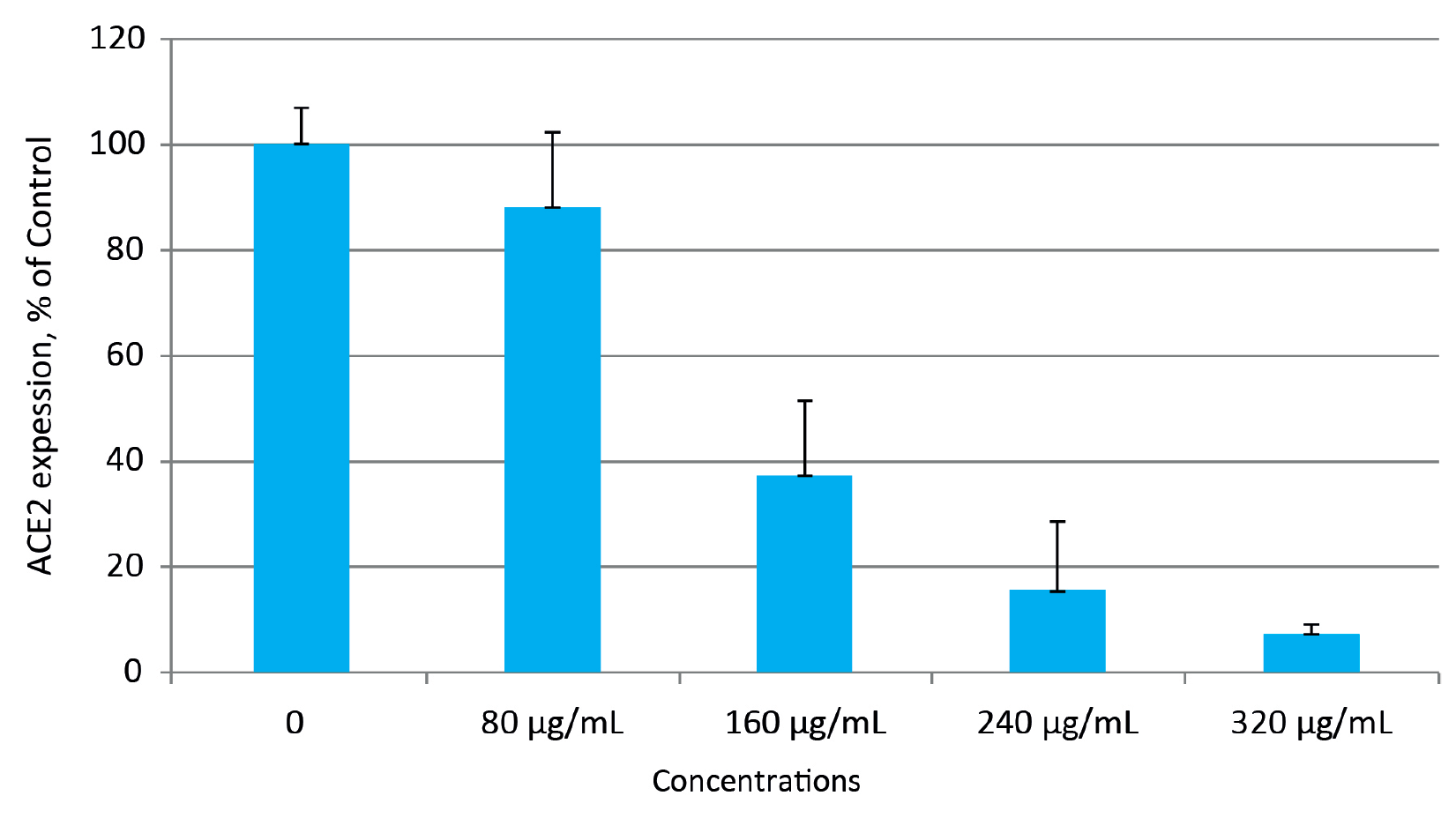 Figure 2: Effects of micronutrient combination on ACE2 receptor expression in human small alveolar epithelial cells. Changes in ACE2 expression are presented as % of control.
Figure 2: Effects of micronutrient combination on ACE2 receptor expression in human small alveolar epithelial cells. Changes in ACE2 expression are presented as % of control.
The effect of specific micronutrient combination on RBD binding to ACE2 receptor
Binding of the RBD sequence of the SARS-CoV-2 Spike protein to its cellular receptor is the necessary step in gaining access to the target cells and for viral infectivity.
We tested the effects of the same specific mixture of active plant components on blocking the interaction of this binding receptor sequence of the SARS-CoV-2 Spike protein with the human ACE2 receptor. In our study we applied a state-of-the-art high sensitivity screening test that can detect inhibitors that block the interaction between the receptor binding domain (RBD) of the viral Spike protein with ACE2 receptor.
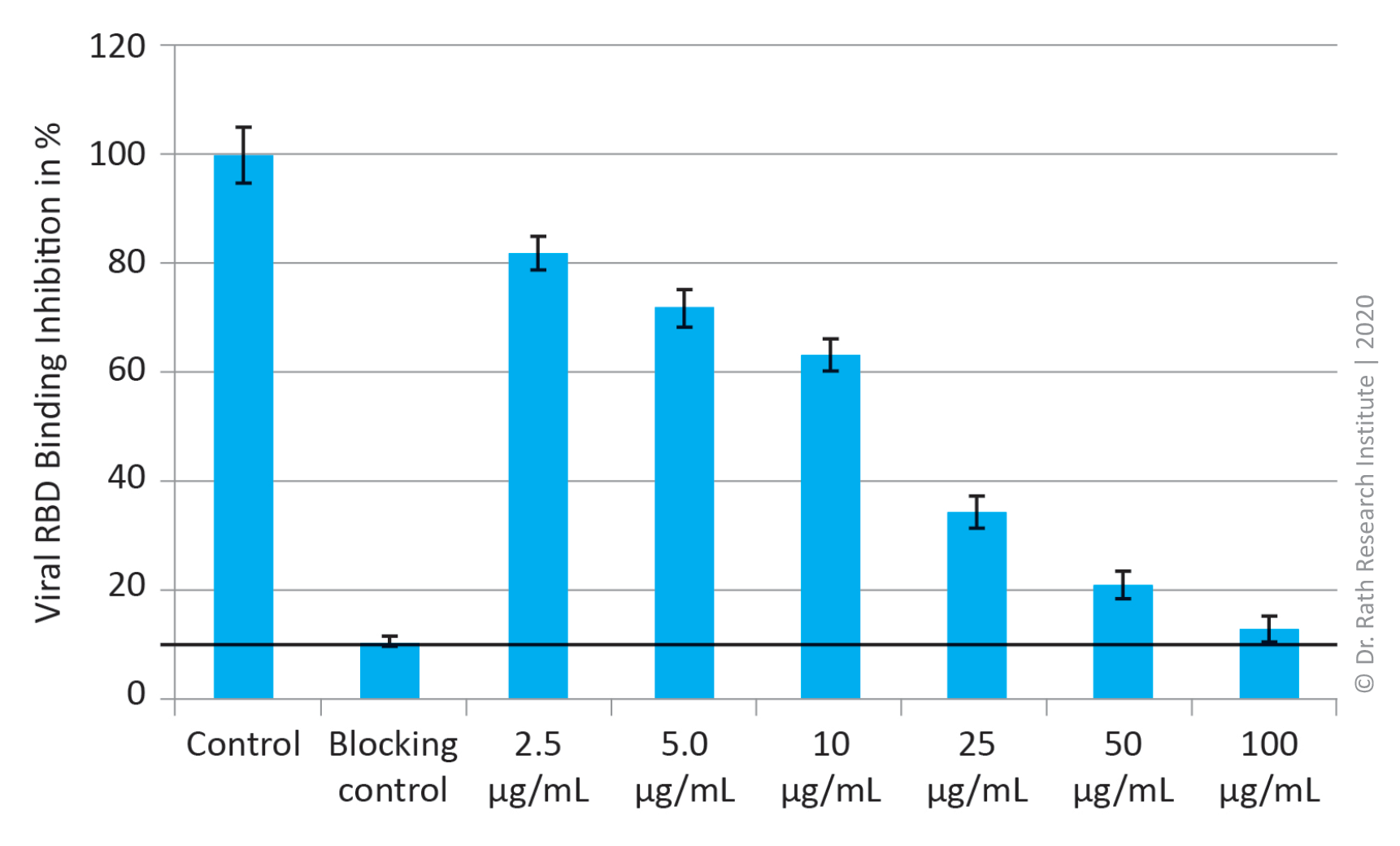 Figure 3: Blocking of SARS-CoV-2 Spike (RDB) binding to ACE2 receptor by specific micronutrient combination in % of control. ‘Blocking control’ = 100% binding inhibition.
Figure 3: Blocking of SARS-CoV-2 Spike (RDB) binding to ACE2 receptor by specific micronutrient combination in % of control. ‘Blocking control’ = 100% binding inhibition.
Figure 3 shows that the test combination of natural plant derived compounds was able to block the attachment of RBD domain on Spike protein of the SARS-CoV-2 virus to its receptor ACE2. This inhibitory effect was concentration dependent and at 100 µg/mL concentration of the mixture it caused 97% binding inhibition. Its strong efficacy in preventing viral binding was observed already at a 40-times lower concentration, i.e. 2.5 µg/mL, which caused about 20% binding inhibition.
DISCUSSION
Significance of our findings
The results presented in this study document that micronutrients can strongly inhibit important cellular mechanisms associated with coronavirus infection.
These data become available at a critical time when the global scientific and medical community is desperately searching for effective solutions to the COVID-19 pandemic.
By applying scientific methodologies and state-of-the- art techniques used in coronavirus research, we have shown that micronutrients can inhibit the binding of RBD on the Spike protein of SARS-CoV-2 to its specific receptor ACE2 by 97%. This means that micronutrients could almost completely prevent viral infectivity. In addition, the same micronutrient composition inhibited the expression of ACE2 receptors on human small alveolar epithelial cells by up to 92%.
Previously, we also reported that another combination of natural compounds including vitamin C, amino acids, plant components, and minerals could significantly decrease the expression of ACE2 receptors on two types of cells mainly targeted by the coronavirus: human lung epithelial and human vascular endothelial cells.9
Another important finding of that study was that inhibition of ACE2 receptors expression was even more pronounced under pro-inflammatory conditions that accompany any viral infection. This would imply that the efficacy of micronutrients would be even more pronounced in clinical conditions and advanced stages of COVID-19, characterized by generalized inflammation and a so-called ‘cytokine storm’.
The present study indicates that specific formulations of plant-derived biologically active compounds can be effective in simultaneously controlling two critical mechanisms involved in the infectivity of the coronavirus.
In light of our findings, it is particularly disappointing that scientific efforts have thus far largely ignored the extensive body of scientific and clinical evidence that substantiate the efficacy of micronutrients in many aspects of viral infections.10, 11, 12, 13
The evidence that natural compounds like micronutrients could be the answer to coronavirus and other pandemics should inspire the scientific
and medical community to embark on a global effort to expand knowledge about the therapeutic value of micronutrients and other natural compounds in the prevention of infectious diseases in general.
Implications of these findings for public health policies
The magnitude of the current pandemic and the dimension of its human and economic costs makes our documentation of the specific value of micronutrients in controlling this pandemic a compelling public health approach.
This is particularly important since all other measures currently developed are either associated with severe side effects or are novel and untested strategies. The cautionary reactions of the international community to the first registered vaccines, which are viral gene- based, testifies to the awareness of the potential risks associated with a population-wide implementation of such technologies.
Based on our study results, a scientifically proven micronutrient-based approach becomes available as an effective – and safe – public health strategy to fight the current pandemic. With a blocking rate of nearly 100%, micronutrients rival the efficacy of any vaccine – yet without any incalculable risks. Moreover, this approach widens the potential health benefits by addressing other critical mechanisms associated with coronavirus infections. These mechanisms include a significant decrease in the availability of viral ‘anchors’ (ACE2) on the surface of human cells and essential support for immune system function.
A micronutrient-based approach also allows the people at large to actively participate in the prevention of coronavirus infections – beyond wearing masks, social distancing, and other defensive measures.
REFERENCES
- Chakraborty I, Maity COVID-19 outbreak: Migration, effects on society, global environment and prevention. Sci Total Environ. 2020; 728: 138882. https://dx.doi.org/10.1016%2Fj.scitotenv.2020.138882
- WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int/. Updated August 13, Accessed August 14, 2020.
- Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020; 382(8): 727-733. doi: 1056/NEJMoa2001017
- Li Structure, Function, and Evolution of Coronavirus Spike Proteins. Annu. Rev. Virol. 2016; 3(1): 237-261. doi:10.1146/annurev-virology-110615-042301
- Li W, Moore MJ, Vasilieva N, et al. Angiotensin- converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003; 426(6965): 450–454. https://doi.org/10.1038/nature02145
- Hofmann H, Pyrc K, van der Hoek L, Geier M, Berkhout B, Pohlmann S. Human coronavirus NL63 employs the severe acute respiratory syndrome coronavirus receptor for cellular entry. Proc. Natl Acad. Sci. USA. 2005; 102(22):7988– 7993. https://doi.org/10.1073/pnas.0409465102
- Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The Spike protein of SARS-CoV—A target for vaccine and therapeutic. Nat. Rev. Microbiol. 2009; 7, 226–236. https://doi.org/10.1038/nrmicro2090
- Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang. MERS-CoV Spike protein: a key target for antivirals. Expert Opin Ther Targets. 2017; 21(2): 131-143. doi:10.1080/14728222.2017.1271415
- Ivanov V, Ivanova S, Niedzwiecki A, Rath M. Effective and save global public health strategy to fight the COVID-19 pandemic: Specific micronutrient combination inhibits Coronavirus cell-entry receptor (ACE2) expression. J Cell Med & Nat. Health, 2020.
- Jariwalla RJ, Roomi MW, Gangapurkar B, Kalinovsky T, Niedzwiecki A, Rath M. Suppression of influenza A virus nuclear antigen production and neuraminidase activity by a nutrient mixture containing ascorbic acid, green tea extract and amino acids. 2007; 31(1): 1-15. doi: 10.1002/biof.5520310101
- Jariwalla R, Gangapurkar B, Pandit A, Kalinovsky T, Niedzwiecki A, Rath M. Micronutrient Cooperation in Suppression of HIV Production in Chronically and Latently Infected Cells. Mol Med 2010; 3(3): 377-85. doi: 10.3892/mmr_00000268.
- Deryabin PG, Lvov DK, Botikov AG, et al. Effects of a nutrient mixture on infectious properties of the highly pathogenic strain of avian influenza virus A/H5N1. Biofactors. 2008; 33(2): 85-97. doi: 10.1002/biof.5520330201.
- Barbour EK, Rayya EG, Shaib H, et al. Alleviation of histopathological effects of avian influenza virus by a specific nutrient synergy. International J Appl Res Vet Med. 2007; 5(1): 9-16.
Source:








